Cell & Gene Therapy Insights is an online, open-access, peer-reviewed journal dedicated to the interdisciplinary exploration and advancement of cell and gene therapy. With a translational focus, we connect innovative research to practical clinical applications, providing valuable insights into one of biotechnology’s fastest-evolving fields.
We address the key challenges and latest developments across advanced therapies, publishing original research articles, expert reviews, commentary, clinical trial reports, and more. Visit the Cell & Gene Therapy Insights Journal page for our complete collection.
Our popular webinar series provides expert-led discussions on important developments and methodologies in the field, designed to support ongoing professional growth.
Explore our specialised channels, including detailed coverage of critical areas like the cell and gene supply chain, for targeted insights into manufacturing, logistics, and regulatory frameworks essential to therapy commercialisation.
Learn more about our journal’s mission and publishing criteria by visiting our aims and scope.
Sign-up for free to gain unlimited access to our extensive library of articles, webinars, podcasts, news, and interviews, and stay at the forefront of cell and gene therapy innovation.
If you’re interested in working with us, from sponsoring articles to webinars and more, view the media kit to find out more.
March 2026
Upcoming webinars

Identifying and addressing key regulatory risks affecting cell therapy programs

Scaling AAV manufacturing across fixed-bed bioreactors: lessons from Novartis (APAC)

New automated, closed system for cell therapy formulation and filling

A scalable CAR-T cell manufacturing workflow producing 100+ doses per batch

New automated, closed system for cell therapy formulation and filling (APAC)

Cell-derived nanoparticles: a new paradigm for CAR-T cell activation and manufacturing
Latest Articles

Safety assays for quality control of ATMPs: addressing mycoplasma detection challenges

Scale-up and process transfer considerations in lentiviral vector manufacturing

Industrializing allogeneic iPSC-derived cell therapies: challenges and solutions

5 ways packaging impacts cell therapy outcomes

Off‑the‑shelf and on‑target: how iPSC‑derived CAR‑T cells could democratize autoimmune cell therapy

Industry Insights: Regulatory dynamics and clinical progress in early 2026

Tailored analytical strategies for AAV aggregation: purpose‑driven method selection for enhanced product development
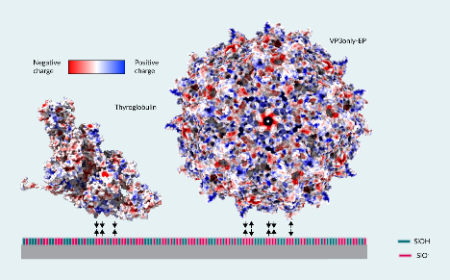
New analytical method for rAAV titer and empty/full ratio determination in cell lysate and conditioned medium