Cell & Gene Therapy Insights is an online, open-access, peer-reviewed journal dedicated to the interdisciplinary exploration and advancement of cell and gene therapy. With a translational focus, we connect innovative research to practical clinical applications, providing valuable insights into one of biotechnology’s fastest-evolving fields.
We address the key challenges and latest developments across advanced therapies, publishing original research articles, expert reviews, commentary, clinical trial reports, and more. Visit the Cell & Gene Therapy Insights Journal page for our complete collection.
Our popular webinar series provides expert-led discussions on important developments and methodologies in the field, designed to support ongoing professional growth.
Explore our specialised channels, including detailed coverage of critical areas like the cell and gene supply chain, for targeted insights into manufacturing, logistics, and regulatory frameworks essential to therapy commercialisation.
Learn more about our journal’s mission and publishing criteria by visiting our aims and scope.
Sign-up for free to gain unlimited access to our extensive library of articles, webinars, podcasts, news, and interviews, and stay at the forefront of cell and gene therapy innovation.
If you’re interested in working with us, from sponsoring articles to webinars and more, view the media kit to find out more.
June 2025
Upcoming webinars

Lessons learned from an end-to-end digitization journey for advanced therapy workflows
Streamline disease modeling and therapeutic discovery with targeted viral vectors

Commercialization of CAR-T therapy in New Zealand: clinical insights & scalable manufacturing
Dual CAR-T therapy targeting solid tumors: insights from intracerebroventricular delivery

Integrating microbubbles into a CAR-T workflow: more doses in a smaller footprint
Automating iPSC culture: scalable workflows
Latest Articles

Mapping the cell and gene therapy landscape: community insights
Industrialization of AAV manufacturing by Xcite® transient and stable production platforms
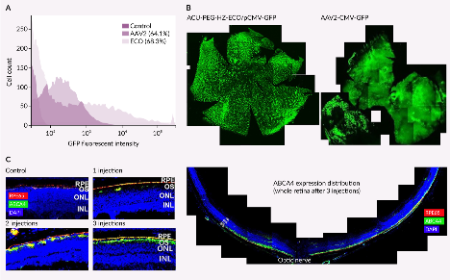
Multifunctional pH-sensitive ionizable lipids for efficient nucleic acid delivery

From CRISPR to DNA writing: expanding the gene editing toolbox
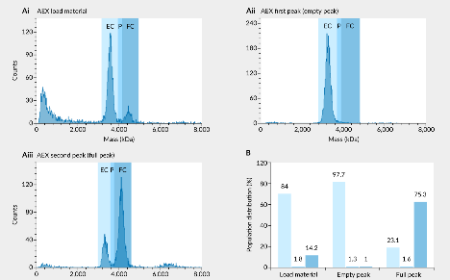
A simplified chromatography method for robust separation of full and empty AAV capsids: a case study with AAV2

From innovation to access: bridging gaps in scalability and cold chain precision logistics for advanced therapies

Bringing cell therapies closer to patients: the role of automation, analytics, and decentralized manufacturing
Enabling decentralized manufacture for cell and gene therapies












