Enhancing non-viral genome editing and manufacturing of T and NK cells for immunotherapies
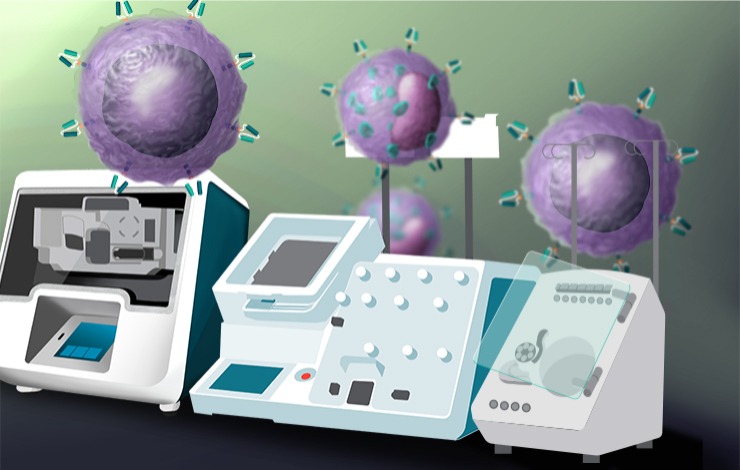
The clinical manufacturing of gene-modified cell-based immunotherapies is complex and labor-intensive. Despite its widespread application, the use of viral vectors for transgene delivery comes with limitations, including high costs, biosafety challenges, and regulatory restrictions. Key current areas of focus for industry include the optimization of non-viral gene delivery methods, and closing and automating cell processing to improve overall safety and cost-effectiveness.
In this webinar, we will discuss tools and protocols for non-viral engineering of CAR-T cells and CAR-NK cells, reviewing data that show efficient gene editing and post-electroporation viability for both cell types. We will discuss a next-generation, universal CAR platform (OmniCAR) developed by Prescient Therapeutics with insights on efforts made to establish closed, GMP-compliant processes for manufacturing in a collaboration with Thermo Fisher Scientific. Speakers will showcase data incorporating a closed end-to-end cell manufacturing workflow utilizing Gibco™ CTS™ Dynacellect Magnetic Cell Separator, Gibco™ CTS™ Xenon™ Electroporation System, Gibco™ CTS™ Rotea™ Counterflow Centrifugation, and ancillary reagents.
Attend this webinar to learn more about:
- Fit-for-purpose, closed, modular instrumentation platforms that can be used to modify and process human NK and T cells for cell therapies
- The potential of human NK cells grown in feeder-free medium in a clinical setting
- How to transition from the small benchtop Invitrogen Neon system to the large-scale CTS Xenon system, suitable for clinical manufacturing
You might also like

Same-section spatial multiomics: a platform for detailed analysis of the solid tumor TME

Quadplex dPCR as a tool to analyze genome integrity and heterogeneity in gene therapy drug products
Precision in production: optimizing monitoring and quality control for high-value plasmids
Advancing cell therapy: innovations in polymer-based encapsulation and delivery



