Cell & Gene Therapy Insights is an online, open-access, peer-reviewed journal dedicated to the interdisciplinary exploration and advancement of cell and gene therapy. With a translational focus, we connect innovative research to practical clinical applications, providing valuable insights into one of biotechnology’s fastest-evolving fields.
We address the key challenges and latest developments across advanced therapies, publishing original research articles, expert reviews, commentary, clinical trial reports, and more. Visit the Cell & Gene Therapy Insights Journal page for our complete collection.
Our popular webinar series provides expert-led discussions on important developments and methodologies in the field, designed to support ongoing professional growth.
Explore our specialised channels, including detailed coverage of critical areas like the cell and gene supply chain, for targeted insights into manufacturing, logistics, and regulatory frameworks essential to therapy commercialisation.
Learn more about our journal’s mission and publishing criteria by visiting our aims and scope.
Sign-up for free to gain unlimited access to our extensive library of articles, webinars, podcasts, news, and interviews, and stay at the forefront of cell and gene therapy innovation.
If you’re interested in working with us, from sponsoring articles to webinars and more, view the media kit to find out more.
February 2026
Upcoming webinars

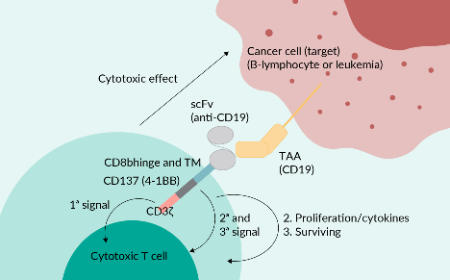
Defining effective antitumor T cell responses to guide next-generation adoptive cell therapies for solid tumors
Steady and stable transfection from lab to launch: enabling seamless viral vector scale-up

Advancing non-viral CAR-NK development: LNP engineering insights and cell therapy regulatory trends

Frozen by design: engineering and automating cryogenic storage to avoid risk and scale-up surprises

dPCR strategies for sensitive quantification and fragment size assessment of residual host cell DNA

Integrating pathology services to improve data quality and trial execution
Latest Articles

From dose quantity to dose quality: elevating rAAV genome integrity as a CMC attribute

Induced pluripotent stem cells and the rise of human-relevant in vitro models: promise, pitfalls, and the path forward

Solid tumors and advanced therapies: why science alone will not deliver the cure

The next era of cell therapy

Next-generation strategies for managing AAV pre-existing immunity: beyond standard companion diagnostics

An analysis of advanced therapy medicinal products regulatory landscape in ICH member countries: opportunities and challenges

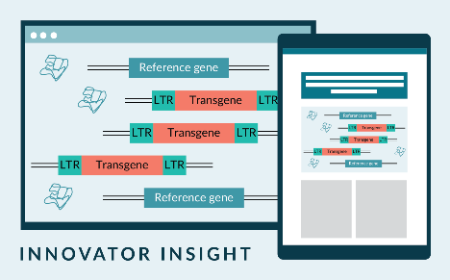
Integration site analysis in engineered T cell therapies: what we measure, what we miss, and what regulators expect

The cryo-sensitivity of NK cells: overcoming post-thaw decline