Optimizing HEK293-based viral vector production, part 2: process and analytics
Cell & Gene Therapy Insights 2024; 10(1), 49
DOI: 10.18609/cgti.2024.010
Published: 7 February 2024
FastFacts
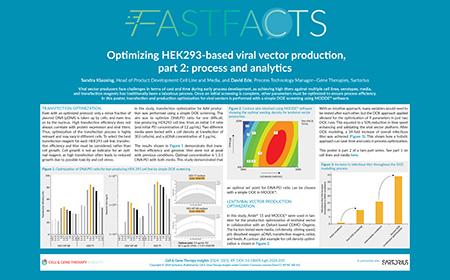 | Watch the video or view the poster to learn how to:
|

