Leveraging rapid sterility testing to advance cell therapy production
Cell & Gene Therapy Insights 2024; 1(4), 609
DOI: 18609/cgti.2024.073
Published: 3 June
FastFacts
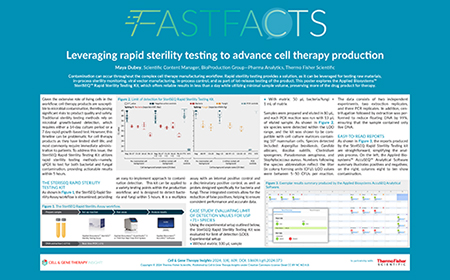 | Watch the video or view the poster to learn about:
|
