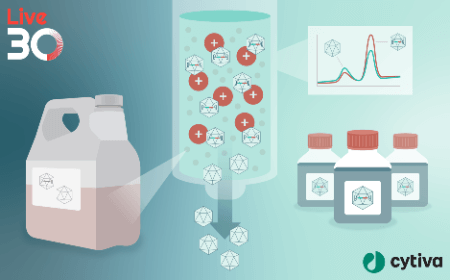
Scalable suspension LVV production platforms for cell and gene therapy
Lentiviral vector is the predominant delivery vehicle used for introducing genetic modifications into cell therapies such as CAR-T and HSC gene therapies. Fueled by recent clinical successes and landmark approvals, this field continues to experience unprecedented pipeline growth and investor interest along with a concommitant growth in demand for Lenti viral vector manufacturing. Traditional processes for Lenti manufacturing rely on lab-scale, open, manual processes that are predominantly adherent-based and difficult to scale up. There is a pressing need for industrialization of these production methods to meet market demand and to ensure successful commercialization of cell and gene therapies.
In this Webinar, Lonza CGT will focus on how we are tackling these challenges through development of an optimized, scalable suspension-based Lenti production platform using our proprietary HEK293T clonal cell line. We will also highlight some of the innovation and process improvements steps that have been implemented to support trailblazers of CGT to generate commercially viable therapies to from concept to patient.
- Lentiviral vector market trends for CGT
- Key manufacturing challenges of Lenti production
- Solutions for developing commercially viable Lenti processes from concept to patient
- Optimized LVV production using a proprietary HEK293T cell line and a scalable, suspension-based production platform
You might also like
Industrialization of AAV manufacturing with Xcite® transient and stable production platforms
Advancing cell therapy: Optimizing the cell culture environment for high-quality manufacture of suspension and adherent cells
Future-proofing gene therapy: scalable AAV solutions and non-viral innovations

Scalable solutions for optimizing lentiviral vector production